In order to make the results commented on above more quantitative, a layered structure of the oxide film was assumed: the metal substrate is covered by a sub- oxide layer, on top of which the HfO, grows whereas a fraction of a monolayer of oxygen atoms is always present at the surface. Possible assignments to the high- binding-energy states are hydroxyl groups and chemi- sorbed oxygen at the surface.20*21 Using a CO2 laser, the change of Cu morphology was monitored during thermal annealing by LEIS. Like Cu/HfO2, the growth mode of Cu on HfSiO4 at 300 K is three-dimensional. Nevertheless, at higher binding energies other components could be detected (cf. viii In the second study, the growth and thermal annealing of Cu on HfSiO4 were studied using PVD, in-situ XPS, in-situ LEIS, and ex-situ AFM. A peak at 531.30 k 0.05 eV binding energy can be observed clearly and was assigned to an oxide state. The analysis of the 0 1s spectra at different oxygen exposures showed more than one component. Previous hafnium work has reported the 4d 5/2 and 4d 3/2 peaks occurring at 211.3 eV and 221.8 eV for hafnium metal 55, 219.1 eV and 229.8 eV for hafnium dioxide (polycrystalline solid) 55.

This behaviour can be explained if it is assumed that part of the suboxides formed at 25 L are further oxidized and transformed into HfO,, whereas the rest remain at the metal/oxide interface. HfO, is not formed until exposures of -10 L, then its intensity increases with exposure and saturation is attained at -500 L.
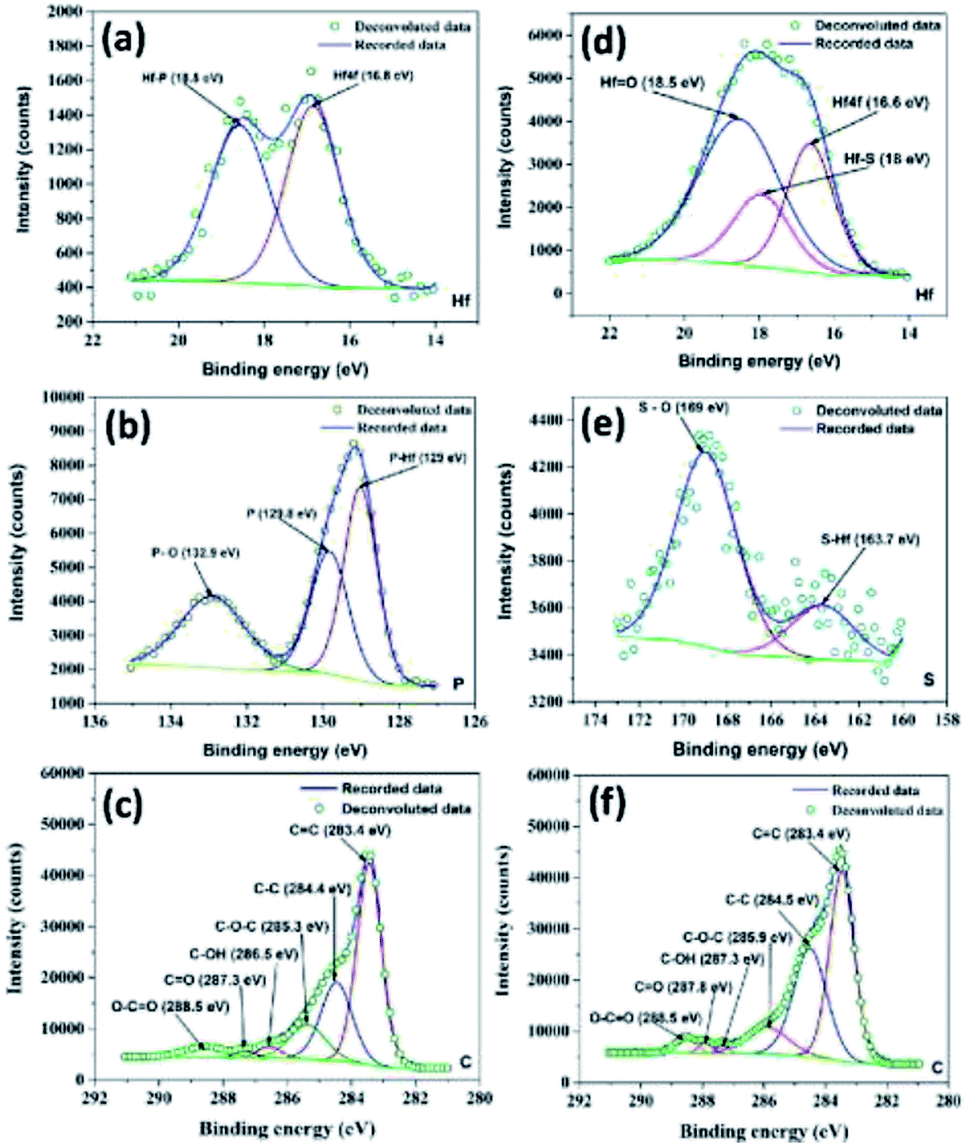
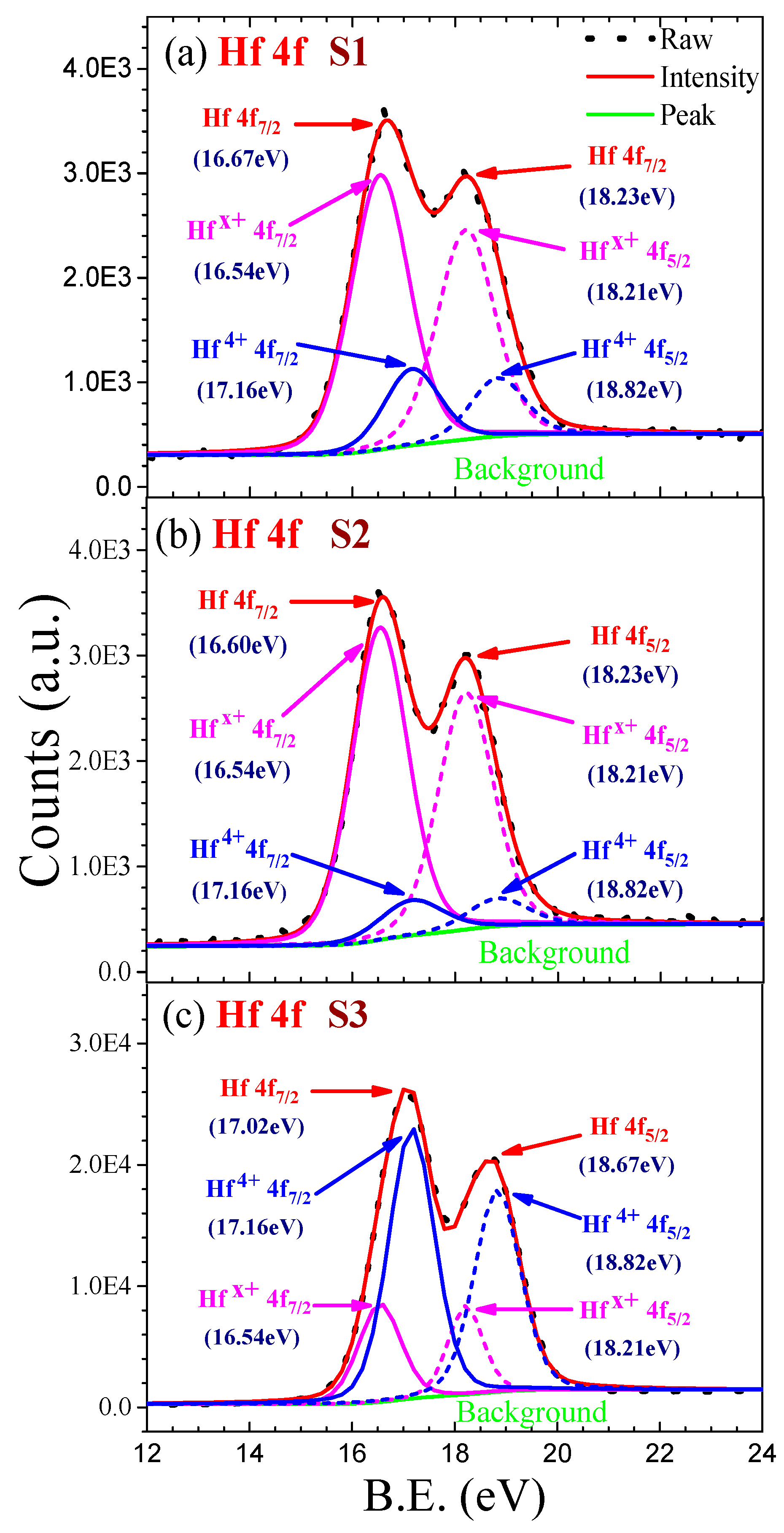
After a chemisorption stage for exposures 500 L. The oxidation kinetics of polycrystalline hafnium (Hf) at room temperature a d low oxygen pressure (Poz - lo-' Ton) has been studied by x-ray photoelectron spectroscopy (XPS). Sanz Departamento de Fisica Aplicada, C-XII, Universidad Aut6noma de Madrid, Cantoblanco, E-28C49, Madrid, Spain 16, 304-308 (1990)Īn XPS Study of the Initial Stages of Oxidation of HafniumĬ.
